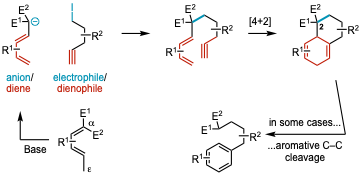We gratefully acknowledge the The NSF (CAREER 1844443; 2019 – 2024) and the NIH (R35 GM137893-01; 2020 – 2025) for funding many aspects of our research program!
Publications:
28. Three-Component cine,ipso-Disubstitution of Nitrocoumarins
Vincent Vedovato, Anghelo J. Gangano, Ion Ghiviriga, and Alexander J. Grenning*
27. Axially Chiral Cannabinoids: Design, Synthesis, and Cannabinoid Receptor Affinity
Sara E. Kearney, Anghelo J. Gangano, Primali V. Navaratne, Daniel G. Barrus, Kyle J. Rehrauer, Terry-Elinor R. Reid, Adrian Roitberg, Ion Ghiviriga, Christopher W. Cunningham*, Thomas Gamage*, and Alexander J. Grenning*
ChemRxiv preprint HERE.
26. Altered methionine metabolism impacts phenylpropanoid production and plant development in Arabidopsis thaliana
Doosan Shin, Veronica C. Perez, Gabriella K. Dickinson, Haohao Zhao, Ru Dai, Breanna Tomiczek, Keun Ho Cho, Ning Zhu, Jin Koh, Alexander Grenning, Jeongim Kim*
25. Vicinal Stereocenters via Asymmetric Allylic Alkylation and Cope rearrangement; A Straightforward Route to Functionally and Stereochemically Rich Heterocycles
Aleksandra Nilova,† Michael D. Mannchen,† Abdias N. Noel, Evgeniy Semenova, and Alexander J. Grenning*
† Equal contribution. These authors reserve the right to list their names first.
24. Diastereoselective Indole-Dearomative Cope Rearrangements by Compounding Minor Driving Forces
Subhadip De, Breanna M. Tomiczek, Yinuo Yang, Kenneth Ko, Ion Ghiviriga, Adrian Roitberg*, and Alexander J. Grenning*
ChemRxiv preprint HERE.
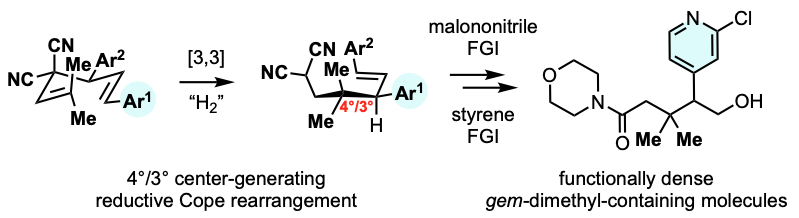
23. Construction of vicinal 4°/3°-carbons via reductive Cope rearrangement.
Kristin M. Sobie, Matthew Albritton, Yinuo Yang, Mariana M. Alves, Adrian Roitberg*, and Alexander J. Grenning*
ChemRxiv preprint HERE.
22. 1,2,4-Trifunctionalized Cyclohexane Synthesis via a Diastereoselective Reductive Cope Rearrangement and Functional Group Interconversion Strategy.
Michael D. Mannchen, Ion Ghiviriga, Khalil A. Abboud, and Alexander J. Grenning*
21. [3,3] Ring Rearrangement of Oxo- or Aza-Bridged Bicyclo[3.2.1]octene-Based 1,5-Dienes.
Evgeniya Semenova, Ouidad Lahtigui, Ion Ghiviriga, and Alexander J. Grenning*
20. Aromatic Cope rearrangements
Breanna M. Tomiczeka and Alexander J. Grenning*
19. Function‐oriented and modular (+/–)‐cis‐pseudoguaianolide synthesis: Discovery of new Nrf2 Activators and NF‐κB Inhibitors
Fabien Emmetiere, Ranjala Ratnayake, Henry A. M. Schares, Katherine F. M. Jones, Emily Bevan-Smith, Hendrik Luesch,* Daniel A. Harki,* and Alexander J. Grenning*
18. Overcoming Kinetic and Thermodynamic Challenges of Classic Cope Rearrangements
Ehsan Fereyduni, Ouidad Lahtigui, Jacob N. Sanders, Breanna M. Tomiczek, Michael D. Mannchen, Roland A. Yu, K. N. Houk,* and Alexander J. Grenning,*
17. Selective ring-rearrangement or ring-closing metathesis of bicyclo[3.2.1]octenes
E. Semenova, O. Lahtigui, S. Scott, M. Albritton, K. A. Abboud, I. Ghiviriga, A. E. Roitberg* and A. Grenning*
16. Functional Group Interconversion of Alkylidenemalononitriles to Primary Alcohols by a Cooperative Redox Operation
F. Emmetiere, A. J. Grenning
15. Axially‐Chiral Cannabinols: A New Platform for Cannabinoid‐Inspired Drug Discovery
Primali V. Navaratne, Jenny L. Wilkerson, Kavindri D. Ranasinghe, Evgeniya Semenova, Jasmine S. Felix, Lance R. McMahon, Adrian Roitberg, Ion Ghiviriga, and Alexander J. Grenning*
14. Diastereoselective Synthesis of 2,3,4-Trisubstituted Tetrahydrofurans via Thermally Reactive 1,5-Diene-tert-butyl Carbonates
Fabien Emmetiere and Alexander J. Grenning
13. Promoting Thermodynamically Unfavorable [3,3] Rearrangements by Chemoselective Reduction
Vertesaljai, P.; Serrano, R.; Mannchen, M. D.; Williams, M.; Semenova, E.; Grenning, A. J.
12. Accessing the decarboxylative allylation–divinylcyclopropane-cycloheptadiene rearrangement from the ketone/aldehyde substrate pool
Schroeder, R.; Grenning, A. J.
11. Controlling, Understanding, and Redirecting the Thermal Rearrangement of 3,3-Dicyano-1,5-enynes
Scott, S. K.; Sanders, J. N.; White, K. E.; Yu, R. A.; Houk, K. N.; Grenning, A. J.
10. Transient [3,3] Cope rearrangement of 3,3-dicyano-1,5-dienes: computational analysis and 2-step synthesis of arylcycloheptanes
Ehsan Fereyduni, Jacob N. Sanders, Gabriel Gonzalez, K. N. Houk*, and Alexander J. Grenning*
9. Tetrahydrobenzochromene Synthesis Enabled by a Deconjugative Alkylation/Tsuji-Saegusa-Ito Oxidation on Knoevenagel Adducts
Navaratne, P. V.; Grenning, A. J.
8. Complex Hydroindoles by an Intramolecular Nitrile-Intercepted Allylic Alkylation Cascade Reaction
Vertesaljai, P.; Ghiviriga, I.; Grenning, A. J.
7. Factors Governing and Application of the Cope Rearrangement of 3,3-Dicyano-1,5-dienes and Related Studies
Fereyduni, E.; Grenning, A. J.
6. An Enyne Cope Rearrangement Enables Polycycloalkane Synthesis from Abundant Starting Materials by a Simple Strategy
Scott, Sarah K. ; Grenning, A. J.
5. Simplifying Complex Scaffold Synthesis: Knoevenagel Adduct Allyl Anions as Easily Generated Multifunctional Reagents
Grenning, A. J.
4. A Deconjugative Alkylation/Diels-Alder Cycloaddition Strategy to Synthesize 2-Substituted Bicyclic Scaffolds
Zhang,; W.; Ghiviriga, I.; Grenning, A. J.
3. Deconjugative Alkylation/ “Pairing” as a Simple Platform for Carbocycle Synthesis
Navaratne, P. V.; Grenning, A. J.
2. Assembly of Terpenoid Cores by a Simple Tunable Strategy
Lahtigui, O.; Emmetiere, F.; Zhang, W.; Jirmo, L.; Toledo-Roy, S.; Hershberger, J. C.; Grenning, A. J.
1. Knoevenagel Adducts as Trimethylenemethane Dipole Surrogates
Vertesaljai, P.; Navaratne, P. V.; Grenning, A. J.